Q1. What are the units of molar heat capacity?
Q2. Tea placed in a closed vessel is an example of
Q3. In Born-Haber cycle, the sum of the enthalpy changes around a cycle is
Q4. Which of the following statements is correct about the process of evaporation of water from an open beaker?
Q5. Which of the following is extensive property?
Q6. The solubility of fluorides is much less as compared to corresponding chlorides
Q7. The increasing order of enthalpy of vaporization of NH3, PH3, and AsH3 is
Q8. Which of the following relations is correct for an adiabatic process?
Q9. Which of the following property is NOT an intensive property?
Q10. Spontaneous processes proceed:
Q11. What is the enthalpy called when one mole of compound is formed under standard conditions from one mole of its constituent elements in their standard state?
Q12. Which of the following is NOT a state function?
Q13. Evaporation of water is:
Q14. Which of the following process is NOT included in phase transformation?
Q15. What happens to the ions, when an ionic compound dissolves in a solvent?
Q16. Born-Haber cycle is used to calculate:
Q17. The sum of internal energy and pressure-volume work is defined as
Q18. The difference between Cp and Cv is equal to
Q19. On which of the following factors the enthalpy change of a reaction is NOT dependent?
Q20. What is the enthalpy called when one mole of compound reacts completely with excess oxygen under standard conditions?
Q21. The process in which no exchange of heat takes place between system and surrounding is known as
Q22. Which of the following process does NOT occur during a chemical reaction?
Q23. What is the change in the entropy of water, When ice melts into water?
Q24. Which of the following compounds requires maximum heat to vaporize?
Q25. Units of entropy change are:
Q26. Heat change of a process in which volume remains constant is given by:
Q27. The internal energy of a system is
Q28. Standard enthalpy of vapourisation
∆vap Ho for water at 100oC is
40.66 kJ mol-1. The internal energy of
vaporization of water at 100oC (in kJmol-1) is:
Q29. For the reaction Hg (l) ---------> Hg (g) , the entropy change will have the sign:
Q30. A system absorbs 500 kJ heat and performs 250 kJ work on the surroundings. The increase in internal energy of the system is:
Q31. Which of the following is true about a spontaneous process?
Q32. The enthalpies of fusion for all reactions are
Q33. In the evaporation of water, the entropy
Q34. For a spontaneous chemical process, the free energy change is
Q35. Which of the following is a state function?
Q36. The enthalpies of all elements in their standard states are
Q37. For the reaction,
A (g) +2B (g) → 2C (g) +3D(g)
the change of enthalpy at 27oC is 19 kCal. The Value of ΔE is
Q38. On increasing the temperature the entropy of the system
Q39. What is the value of the enthalpy of neutralization for all strong acids and bases?
Q40. What does term specific heat implies?
 is the enthalpy change when one mole of compound is formed under standard conditions from one mole of its constituent elements in their standard state.
is the enthalpy change when one mole of compound is formed under standard conditions from one mole of its constituent elements in their standard state. of combustion (or the heat of combustion) is the enthalpy change when one mole of compound reacts completely with excess oxygen under standard conditions.
of combustion (or the heat of combustion) is the enthalpy change when one mole of compound reacts completely with excess oxygen under standard conditions.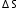 =
= 



 < 0.
< 0. , the value of
, the value of 
Comments
Post a Comment